Page 68 - Summer2020
P. 68
ADAPTED EARS OF TERRESTRIAL MAMMALS
Is the Inner Ear of the Tiger Adapted to React Rapidly to Low Frequencies?
Although the middle ear specialization observed in golden moles is evident, even in gross anatomical terms, some adaptations are more subtle and are recognized only in behaviors or physiologies buried deep in otherwise generalist phenotypes. Discovering those traits can be challenging and are frequently revealed, as with many sci- entific discoveries, through serendipity. Such was the case when an unusual feature in the auditory phenotype of the tiger (Panthera tigris) was discovered. Its discovery sug- gested that the tiger may be best thought of as an auditory specialist, a question that we address in this section. When thinking about use of the term “specialist,” we generally refer to any trait that differentiates the animal’s perfor- mance from closely related organisms. If, for example, the shape of an animal’s audiogram breaks radically from that of other related taxa and is beneficial from an ethological perspective, the animal can be thought of as specialized in that specific trait.
We start, therefore, by pointing out that many elements of the tiger auditory phenotype are typical of auditory gen- eralists. For example, overall sensitivity to acoustic stimuli in tigers is similar to that of other cat (felid) species, and, although the high-frequency limit is lower than that of smaller felids, the shapes of their sensitivity curves are also similar (Walsh et al., 2011). It is also likely that the low- frequency limit of hearing is lower than that of smaller cats, a prediction based on structure-based scaling of the middle ear (Huang et al., 2000).
Although the middle ear transforms sound energy col- lected from the outer ear and transfers it to the inner ear, the cochlea functions as a frequency-analyzing system that is commonly modeled as an array of bandpass fil- ters reflecting the resonant properties of the basilar membrane (BM; Figure 1B), the vibratory membrane supporting the organ of Corti, on which sit the sensory cells for hearing. The resonant properties of the BM reflect the continuously changing mass and stiffness of the membrane along the length of the cochlea (Figure
1A), which increases and decreases, respectively, along a basoapical gradient. This system, coupled with the voltage-dependent motility of a subset of cochlear sen- sory cells (outer hair cells) that amplifies BM motion and produces sharp inner ear filters (Brownell, 2017), can be
thought of as the gateway to audition. The output of these filters determines precisely what a species can detect in the soundscape, a property that, to a large extent, deter- mines if a selective advantage can be gained by enhanced sensitivity to a particular frequency band or by a mecha- nism that enhances frequency selectivity, for example.
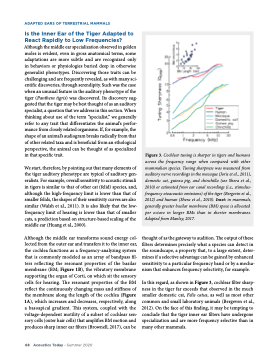
In this regard, as shown in Figure 3, cochlear filter sharp- ness in the tiger far exceeds that observed in the much smaller domestic cat, Felis catus, as well as most other common and small laboratory animals (Bergeven et al., 2012). On the face of this finding, it may be tempting to conclude that the tiger inner ear filters have undergone specialization and are more frequency selective than in many other mammals.
Figure 3. Cochlear tuning is sharper in tigers and humans across the frequency range when compared with other mammalian species. Tuning sharpness was measured from auditory nerve recordings in the macaque (Joris et al., 2011), domestic cat, guinea pig, and chinchilla (see Shera et al., 2010) or estimated from ear canal recordings (i.e., stimulus- frequency otoacoustic emissions) of the tiger (Bergevin et al., 2012) and human (Shera et al., 2010). Inset: in mammals, generally greater basilar membrane (BM) space is allocated per octave in longer BMs than in shorter membranes.
Adapted from Manley, 2017.
68 Acoustics Today • Summer 2020